Trending News
GOVERNMENT WARNS AGAINST USE OF OZEMPIC FOR WEIGHT LOSS

The Pharmacy and Poisons Board (PPB), has issued a safety alert targeting the growing and dangerous trend of using diabetes medication for weight loss.
This warning comes amidst a surge in social media promotions by influencers and the increasing availability of these prescription-only injections in clinics and aesthetic spas across the country.
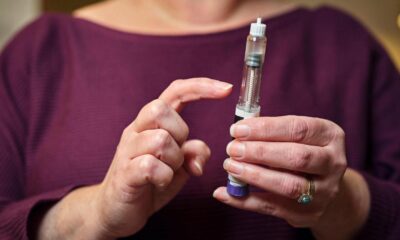
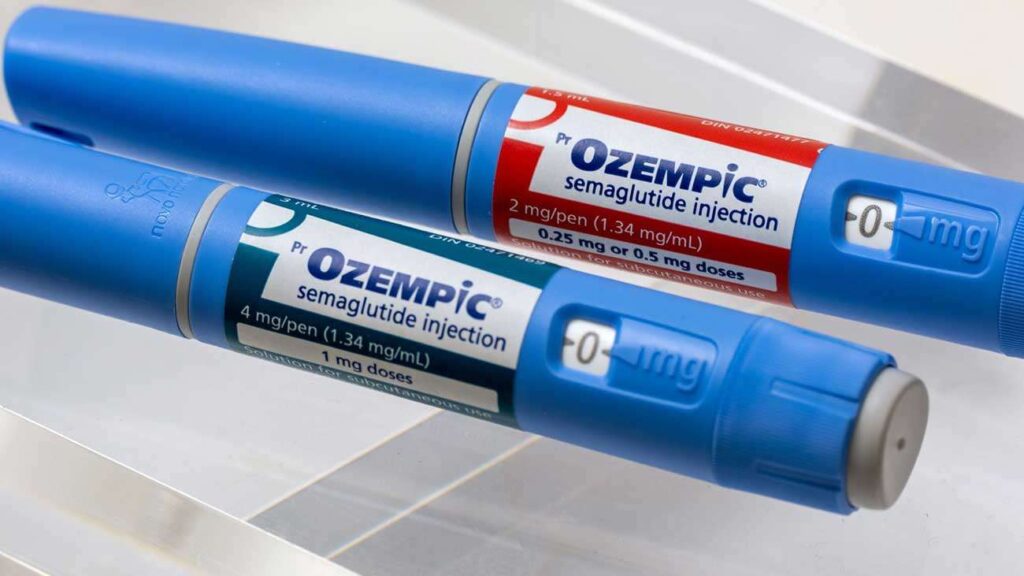
The drug in question, Semaglutide popularly known by brand names like Ozempic has been hailed online as a quick-fix solution to shed weight. Influencers and celebrities have been praising its effects, often without disclosing the significant health risks or its intended medical purpose. This hype has led to a high demand, with some clinics now openly offering it “off-label” for cosmetic weight loss, sometimes without proper medical supervision or prescription.
However, the PPB is now sounding the alarm. In an official statement, the Board clarified that “Semaglutide is a Prescription-Only Medicine and its unsupervised or off-label use may result in serious health outcomes,” said Dr Fred Siyoi, Pharmacy and Poisons Board Chief Executive Officer,
Approved strictly for controlling Type 2 diabetes in Kenya, using it without supervision can lead to severe side effects, including low blood sugar (hypoglycemia), eye conditions, acid (gastroesophageal) reflux disease, and intestinal obstruction, which can range from mild to severe.
Kenyans who have experienced adverse effects or have concerns about the quality of a product are urged to report it immediately, “The public is advised against the off-label use of the medicines and encouraged to report any suspected side effects and poor quality products through the official reporting channels of the Pharmacy and Poisons Board (PPB).”